Trial oversight
This was a phase IIa double-blind, placebo-controlled, randomized clinical trial. The sponsor, Small Pharma (now Cybin UK), designed and supported the trial and provided a proprietary pharmaceutical-grade formulation of DMT fumarate (SPL026), which was analyzed for stability and purity. The trial evaluated the efficacy and safety of DMT, administered as a single or double IV dose, to participants with MDD. The trial was conducted at Hammersmith Medicines Research (HMR), MAC Clinical Research (MAC) and Imperial College London Hammersmith Campus under their respective standard operating procedures.
HMR supervised the trial’s execution, and independent site raters conducted blinded assessments of participants using validated tools, ensuring raters were unaware of trial-group assignments. Data management and analysis were performed by HMR, reviewed by the sponsor and supplemented with post hoc statistical analyses conducted by the sponsor. Small Pharma, as Sponsor, had full oversight of the clinical trial. Data were collected via an electronic case report form using the Medrio EDC system (latest version at the time of use, 2020–2022).
The trial adhered to International Council for Harmonization Good Clinical Practice guidelines and the ethical principles of the Declaration of Helsinki. The trial protocol was reviewed and approved by the UK Medicines and Healthcare products Regulatory Agency and the London–Brent Research Ethics Committee. All participants provided written informed consent. The trial is registered on ClinicalTrials.gov (NCT04673383) and ISRCTN (ISRCTN63465876), where the clinical study report synopsis is available.
Participants
Adults aged 18 years or over with a diagnosis of moderate-to-severe MDD and a history of at least two prior unsuccessful treatment attempts (pharmacological or/and psychotherapeutic) were recruited formally through databases held by the clinical trial sites, informally through social media, and through other sources, which directed participants to a recruitment website. The main exclusion criteria were an immediate family or personal history of psychosis, medically significant health conditions that make a person unsuitable to participate in the trial (as assessed by a physician), a history of serious suicide attempts, a positive pregnancy test, the use of serotonergic psychedelic drugs in the previous 6 months or the suspected or known presence of a pre-existing psychiatric condition (for example, borderline personality disorder) that could jeopardize rapport between the participant and their two trial therapists in light of the protocol-defined limits of available therapist support. Additional details about the trial exclusion criteria are provided in Supplementary Methods (Inclusion and exclusion criteria).
Volunteers initiated contact by emailing the recruitment coordinator after hearing about the trial. Most of the recruited participants referred themselves and some were recruited through patient databases. Candidates were sent a participant information sheet and invited to a telephone screening. Initial assessments of eligibility and suitability were performed by means of a video call. Confirmation of a diagnosis of depression and medical history were obtained from the participant’s general physician. Eligible participants then underwent face-to-face physical and mental health assessments (including the Mini-International Neuropsychiatric Interview) with a trial physician, psychiatrist and therapist (visit 1). Assessment with the 17-item Hamilton Depression Scale (HAM-D-17) was done by the site psychiatrist; a score of at least 17 (indicating moderate-to-severe MDD) on a scale that ranges from 0 to 52, with higher scores indicating greater depression, was required for trial enrollment. Any participants taking AD medication at screening discontinued the use before starting the trial (with the support of a study psychiatrist), with full discontinuation occurring at least 2 weeks before starting a trial medication.
Participants were financially compensated for their time.
The tables use the term sex rather than gender, but this was determined by self-reporting and may reflect gender identity rather than sex assigned at birth. Sex-based/gender-based analyses have not been performed due to overall low sample size. The race/ethnicity categories used followed the FDA’s recommendations which are as follows: American Indian or Alaska Native; Asian; Black or African American; Native Hawaiian or other Pacific Islander; and White. Participants were allowed to self-report and could also designate themselves as multiracial. Ethnicity was separated from race, with the categories—Hispanic/Latino or not Hispanic/Latino. These categories were not used for any inferences or analyses.
Trial design
The sponsor-initiated trial reported here constitutes part B of a larger study investigating the safety, tolerability and efficacy of DMT fumarate in healthy participants (part A) and participants with MDD (part B). Findings from part A, conducted in healthy participants, have been published previously10,20 and the remainder of this manuscript refers to part B only. Enrolled participants with MDD followed a two-stage, double-blind, randomized, placebo-controlled design, with a planned enrollment of 28 to 36 participants (34 were randomized).
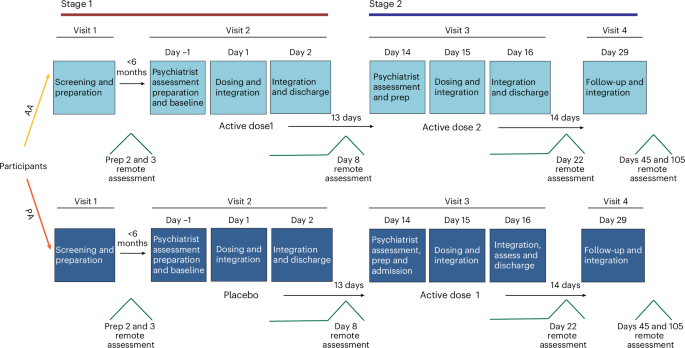
Participants received up to two single IV doses of DMT fumarate or placebo accompanied by a time-limited, relational psychotherapeutic framework focused on psychological flexibility (Supplementary Methods (Therapeutic support)). In stage 1, participant numbers were allocated to blinded treatments (active or placebo) according to a 1:1 randomization schedule prepared by an independent HMR statistician, using SAS statistical analysis software. Two weeks later, in stage 2, participants received a second, open label, dose of DMT, either as their first exposure for those who received placebo in stage 1, defined as PA arm (PA group) or as a second dose for those who received DMT in stage 1, defined as AA arm (AA group). Participants who declined a second dose or were deemed unsuitable for further dosing by the study psychiatrist proceeded to follow-up assessments after their first dose (defined as A- arm). Due to the noticeable psychedelic effects of DMT fumarate, it was likely that both participants and investigators could determine whether a participant had received the active drug or placebo. Despite this, stage 1 of the study adhered to double-blind principles, while stage 2 was conducted as an open-label phase. More information on blinding is provided in the Supplementary Methods (Blinding).
The dosing regimen was based on data from part A and selected for safety and tolerability. The dose administered was 21.5-mg DMT fumarate, infused intravenously over 10 min in two phases—6 mg over the first 5 min, followed by 15.5 mg over the subsequent 5 min (see refs. 10,20 for details). Details of the screening process can be found in Supplementary Methods (Trial visits).
Dosing took place in a controlled inpatient setting, with participants admitted to the ward 1 day prior and discharged the following morning. Preparation involved a 90-min session with two therapists the day before dosing, including orientation to the setting, a guided visualization to promote relaxation and interpersonal safety, and discussion of participant expectations. On dosing day, an additional brief session supported emotional readiness and anticipatory anxiety. Participants received IV DMT in a nonclinical, softly lit room designed to promote calm and containment. They wore eyeshades and noise-canceling headphones and listened to a specially curated, nonverbal ambient music playlist produced by M. Cooper. The music was tailored to follow the experiential trajectory of the DMT session, expansive and immersive during the peak, then gently grounding toward the end. Two therapists were present throughout the session to provide silent, attentive support, while a study psychiatrist remained on site.
Integration was delivered using a relational and participant-led framework developed from previous psilocybin trials. This approach encourages open exploration of emotional, embodied and transpersonal elements of the experience, without prematurely fixing meaning. Integration sessions were conducted in person on day 1 and day 2 postdose, and again on day 15, with therapists helping participants reflect on the psychedelic material and explore how insights could be applied to daily life. Additional follow-up check-ins supported ongoing integration and monitored well-being. Full procedural details for trial visits and the therapeutic framework are provided in the Supplementary Methods (Trial visits and Therapeutic support).
Remote follow-up assessments were conducted 1-week postdose, an in-person visit at day 29 and remote visits 1 month, and 3 months after the final dose. MADRS was assessed (using the SIGMA guide) by a trained independent site rater who was not present during dosing. An overview of the trial is shown in Fig. 1.
In addition to the main trial, an exploratory out-of-study 6-month follow-up visit was performed to monitor long-term outcomes (Supplementary Methods (Trial visits)).
Power calculations
The sample size calculation was based on a two-sided, two-sample t test with equal variance at a significance level of 0.05 and a 1:1 allocation ratio, using data from ref. 30. A sample size of 28 to 36 participants provided 80% to 90% power to detect a 12.5-point mean difference in MADRS score change from baseline.
Efficacy outcomes
The primary outcome of this trial was the change from baseline in the MADRS31 score at 2 weeks (day 14; ±2 days) after the first dose. Secondary efficacy measures included MADRS scores assessed at 1 week (day 8; ±1 day) after the first dose and at 1, 2, 4 and 12 weeks after the second dose (day 22; ±1 day, 29; ±2 days, 45; ±2 days and 105; ±5 days). Additional psychometric tools included the BDI-II32 and the Spielberger’s STAI-T33. These instruments were used to evaluate broader aspects of mood and anxiety that complement the primary depression-related outcome. Participants who did not receive a second dose but remained in the study completed the same follow-up assessments as other participants. The BDI-II and STAI-T were administered in paper form for self-assessment. MADRS was completed on a paper form by an independent site rater after the SIGMA guide34. Instructions were provided at the top of the first page of each questionnaire. The MEQ35, as well as other subjective measures like Ego Dissolution Inventory36, Emotional Breakthrough Inventory37, 11 Dimensions Altered States of Consciousness38, Psychological Insights Scale39, Challenging Experience Questionnaire40 and Individual-Rated Visual Analog Scales, were used to measure the subjective effects of DMT. These subjective scores were also examined to see if they could predict subsequent AD effects. We also examined whether elements of psychedelic preparation (therapeutic rapport, intentions, set and setting) influenced AD response at 2 weeks using the psychedelic predictor scale41. Exploratory analyses also assessed for possible influence of AD washout and prior psychedelic use on AD effects at 2 weeks. A completed list of the measures collected in this trial, including the ones not reported here, can be found in Supplementary Methods (Complete list of collected measures) and Supplementary Table 1.
Safety and tolerability
Adverse events were recorded throughout the study and categorized by severity (mild, moderate, severe) and relationship to treatment (possibly or unlikely related). TEAEs were those occurring postrandomization—from dosing on day 1 to day 14 (stage 1) or from day 15 to day 105 (stage 2). Day 15 TEAEs occurring before dosing on day 15 were excluded from summary tables. Serious adverse events were defined per regulatory guidelines and managed by the sponsor. Additional details on adverse event collection are provided in the Supplementary Methods (Additional adverse event collection methods). Safety monitoring included blood pressure and heart rate (predose and at 12, 60 and 240 min after dosing), ECGs (predose and at 180 min after dosing) and lab tests. Tolerability was assessed postdose by asking if participants regretted the experience. Injection site assessments recorded pain, tenderness, erythema or induration.
Statistical analyses
The primary endpoint was the change in MADRS score between the AA and PA groups from baseline to week 2, with the week 1 change as a secondary outcome, assessed using two-sample t tests during the blinded phase (stage 1). Secondary endpoints, including sustained effects of DMT and the potential benefit of an additional dose, were analyzed using an MMRM. The first MMRM analysis compared MADRS score changes at 1 week (day 22), 2 weeks (day 29), 1 month (day 45) and 3 months (day 105) during the open-label phase (stage 2) between participants who received a single DMT dose (PA group) and those who received two doses (AA group). Additional MMRM analyses explored within-participant changes in MADRS scores after each dose in the AA group and assessed whether the timing of the first DMT dose influenced clinical outcomes (Supplementary Results (‘Secondary analysis—MADRS score comparison between PA and AA groups in stage’ to ‘Supplementary analysis—MADRS remission rates’)). Response and remission rates, defined as >50% reduction in MADRS score and an MADRS score ≤10, respectively, were analyzed using logistic regression adjusted for baseline scores. Changes in BDI-II and STAI-T scores and MADRS scores at the 6-month follow-up (day 224) were summarized descriptively. Moderation effects of the acute psychedelic experience, measured by MEQ scores, on depression severity were explored (Supplementary Results (Supplementary materials—acute measures and moderation effects of Mystical Experience (MEQ) scores and related measures of the acute psychedelic experience) and Supplementary Fig. 2). In response to reviewer feedback, a worst-case scenario sensitivity analysis was conducted to evaluate robustness against potential nonrandom missingness of data in the open-label phase. These results are presented in Supplementary Results (Imputation analyses for handling missing data) and confirmed the results of the main analyses. All analyses, excluding the moderation effects, were done after the statistical analysis plan had been signed and after the database had been locked. The statistical analyses were done by an HMR statistician, using SAS (version 9.4; moderation effects were analyzed by Imperial College using R, the latest version at the time of analysis).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
link