Study design
We conducted a two-arm, parallel-group, assessor-blinded, randomized controlled trial (RCT). This trial was designed to evaluate the efficacy of a personalized, theory-based psychological intervention compared to treatment-as-usual (TAU) on medication adherence in outpatients with depression. The intervention was delivered over a 12-week period, comprising an initial predictive phase (Weeks 0–4) and a subsequent Process-Based Phase (Weeks 5–12). The trial was conducted at the Department of Psychiatry and Psychology of a tertiary general hospital in Guangdong Province, China.
Participants
Participants were recruited via convenience sampling from first-visit outpatients at the study site. All participants were receiving routine multidisciplinary psychiatric care. After randomization, participants were included in only one study arm (intervention or TAU). Researchers collecting outcome assessments were blinded to the participants’ group allocation.
This study was conducted in accordance with the ethical principles of the Declaration of Helsinki and was approved by the Ethics Committee of Southern Medical University (Approval Nan Yi Lun Shen 2021 No.011). Prior to enrollment, all participants (and their parents or legal guardians, where applicable) received a comprehensive explanation of the study and provided written informed consent.
To ensure confidentiality, all participant data were de-identified and stored under a unique code, with access restricted to the principal investigators. The authors declare no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Eligibility criteria
Inclusion criteria were: (1) Outpatients with a clinician-confirmed diagnosis of a depressive episode according to DSM-5 criteria; (2) A score of ≥53 on the Self-Rating Depression Scale (SDS); (3) Minimum education level of primary school; (4) Access to a telephone for follow-up purposes.
Exclusion criteria were: (1) Presence of organic brain diseases; (2) Current psychotic symptoms; (3) Diagnosis of organic depressive disorders; (4) Severe comorbid somatic diseases that could interfere with participation; (5) Substance abuse or dependence; (6) A history of severe drug allergy.
Eligibility was confirmed by a study psychiatrist based on medical record review and clinical assessment.
Randomization and blinding
Eligible participants were randomly allocated (1:1) to the intervention or TAU control group after the completion of all baseline assessments (T1).
An independent statistician generated the randomization schedule (block sizes of 4 and 6), prepared by an independent statistician. Allocation was stratified by sex to ensure balance between groups. Allocation concealment was ensured using sequentially numbered, opaque, sealed envelopes. These envelopes were stored in a locked location and were opened sequentially by a research coordinator only after a participant’s baseline data had been collected.
Due to the nature of the psychological intervention, participants and the intervention providers could not be blinded to group assignment. However, outcome assessors, the data management team, and the trial statistician were blinded to group allocation for the duration of the study. Participants were instructed not to reveal their assignment to assessors.
Interventions
Experimental intervention (personalized, theory-based psychological intervention)
The experimental intervention was a 12-week, multi-component program grounded in behavioral change theories. It was delivered across eight individualized, 30-min sessions. The intervention was structured in two phases:
Predictive phase (weeks 0–4)
The baseline PSPMA assessment guided the application of tailored strategies across five key domains: Autonomy Enhancement, Perceived Necessity, Medication Beliefs, Family Support, and Self-Efficacy. This included psychoeducation, MI, and collaborative problem-solving.
Process-based phase (weeks 5–12)
Intervention strategies were dynamically adapted weekly based on the participant’s assessed Stage of Change (Precontemplation, Contemplation, Preparation, Action, Maintenance). Focus shifted from building motivation in early stages to reinforcing behavior and preventing relapse in later stages. The intervention was delivered by trained psychological research staff under the supervision of a licensed clinical psychologist.
Control intervention (treatment-as-usual—TAU)
Participants allocated to the TAU group received standard outpatient care, which included regular psychiatric consultations, medication management, and general advice. They did not receive the structured, theory-based psychological intervention provided to the experimental group.
Outcome measures
Assessments were conducted at baseline (T1), week 2 (T2), week 4 (T3), week 8 (T4), and week 12 (T5) as detailed in Table 1. Data were collected through on-site paper-based questionnaires, telephone follow-ups, and electronic medical record reviews.
Primary outcome
The primary outcomes of this study were assessed using two validated instruments to capture changes in depressive symptomatology and medication adherence behavior:
Self-Rating Depression Scale (SDS)
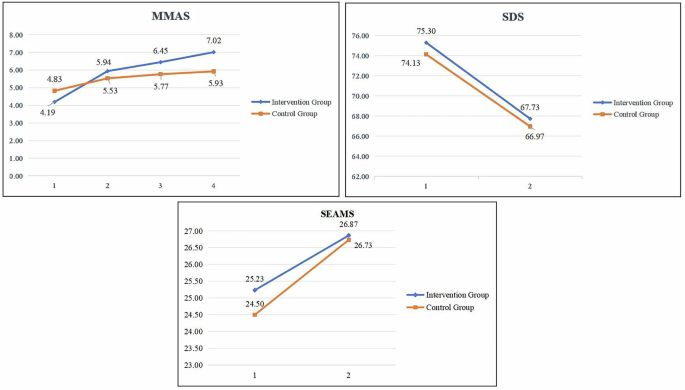
The SDS is a widely used 20-item self-report measure that assesses the affective, cognitive, and somatic dimensions of depression. Each item is scored on a 4-point Likert scale. The total score ranges from 20 to 80, with higher scores indicating more severe depressive symptoms. This scale was selected as a primary endpoint to directly evaluate the intervention’s impact on the core clinical manifestation of depression.
Morisky Medication Adherence Scale (MMAS)
The MMAS is an 8-item self-report questionnaire designed to measure adherence behaviors and attitudes toward psychotropic medications. The MMAS was chosen as a co-primary outcome because improving adherence is the central objective of the proposed intervention.
Secondary outcomes
Secondary outcomes were employed to explore the intervention’s effects on underlying mechanisms and predictive factors related to adherence:
Predictive scale for psychotropic medication adherence (PSPMA)
This scale was used to identify patients at high risk of non-adherence at baseline and to monitor changes in predictive risk factors (e.g., beliefs about medication, perceived self-efficacy) throughout the study period.
Self-efficacy for Appropriate Medication Use Scale (SEAMS)
The SEAMS is a 13-item instrument that assesses a patient’s confidence in their ability to manage medication use under various challenging situations. Responses are scored on a 3-point scale, and higher total scores reflect greater medication self-efficacy. This measure was included to evaluate whether the intervention effectively enhanced this critical psychological mechanism, which is theorized to mediate long-term adherence behavior.
Sample size
The sample size was determined a priori for the marginal model (GEE) analysis of the primary continuous outcome. The sample size calculation was based on the Zhang–Ahn formula for marginal models. Key assumptions included a medium effect size (Cohen’s d = 0.50) at 12 weeks, an exchangeable working correlation structure with an intra-class correlation coefficient (ICC) of ρ = 0.20, and three repeated measurements (baseline, 6-week, and 12-week). With α = 0.05 (two-sided) and power = 0.80, a total of 66 participants (33 per group) were required. Accounting for an estimated 20% attrition over 12 weeks, we aimed to recruit 80 participants (40 per group).
Statistical analysis
Analyses were performed using SPSS 28 (IBM Corp.). Two-tailed α was set at 0.05; Bonferroni–Holm correction was applied when multiple contrasts were tested.
Continuous repeated measures
Medication adherence (MMAS-8), depressive symptoms (SDS), self-efficacy (SEAMS), and PSPMA sub-scores were analyzed with generalized estimating equations (GEE, unstructured correlation, robust SEs). The model included the fixed effects of Group, Time (as a categorical factor), and their Group × Time interaction. Effect sizes are Cohen’s d from model-implied marginal means. Simple contrasts used the Bonferroni–Holm adjustment.
Ordinal outcomes
Stage of Change and Medication Effectiveness Rating were analyzed with ordinal GEE (logit link, unstructured correlation); proportional-odds assumptions were met (Brant test p ≥ 0.08).
Robustness
All models were re-estimated with AR(1) correlation, and after excluding two participants who switched to mania, significance and effect magnitudes were unchanged. VIF < 1.9 excluded multicollinearity.
link